Spravato® Treatment Resistant Depression Clinic

Have other depression treatments failed? Start your journey to relief with SPRAVATO®
Major Depressive Disorder (MDD) is one of the most common mental health disorders in the United States. Treatments such as psychotherapy, medication management, and lifestyle changes may help relieve symptoms. However, other individuals may have Treatment Depression (TRD) and continue to experience symptoms such as persistent feelings of sadness, sleep disturbances, low energy, and thoughts of death or suicide despite trying multiple treatments.
Explore new hope with treatment resistant depression options at our state-of-the-art SPRAVATO treatment program:
SPRAVATO® (esketamine nasal spray) is a new depression medication and an innovative nasal spray treatment for depression that hasn’t improved with other medications. At the Rochester Center for Behavioral Medicine, we provide safe, in-office SPRAVATO® care with the support of an experienced team.
Our medical director, Dr. Joel Young, was a primary investigator during the SPRAVATO® clinical trial. RCBM has 30+ years of experience helping patients find relief from depression, anxiety, and more, guided by Dr. Joel Young and our trusted clinical team.
Should I choose SPRAVATO® or TMS?
What is SPRAVATO®?
SPRAVATO® is the brand name of esketamine. Esketamine is made from a drug called ketamine, an anesthetic that has also been used for many years to treat depression. Because esketamine is more potent, it can be used at a lower dose and has fewer side effects. SPRAVATO® is a unique treatment for individuals who have treatment resistant depression. It involves the antidepressant esketamine delivered in a nasal spray not responded to oral medications.
While SPRAVATO® and ketamine are chemically related, SPRAVATO® is not the same as ketamine. Only SPRAVATO® has undergone extensive controlled clinical trials that informed the FDA approval of the medicine for use in adults with TRD and to treat depressive symptoms in adults with MDD with acute suicidal ideation or behavior.
How It Works
SPRAVATO® (esketamine) belongs to a class of medications called N-methyl-D-aspartate (NMDA) receptor antagonists. It works differently than conventional antidepressants, which alter concentrations of various neurotransmitters, including dopamine, serotonin, and norepinephrine. By targeting NMDA receptors in the brain, SPRAVATO® boosts glutamate levels, the brain’s most prevalent chemical messenger. By increasing glutamate activity, SPRAVATO® enhances communication between brain cells, helping to stabilize mood and improve cognitive function. This results in a greater reach and more substantial impact on brain cells with a single dose compared to conventional antidepressants, potentially leading to faster and more effective relief of depressive symptoms.
Benefits and Safety
Effectiveness in Treatment Resistant Depression
SPRAVATO® has been specifically effective for individuals with treatment resistant depression. Studies have shown that a significant percentage of these patients experience significant improvement after just a few sessions, offering hope to those who have struggled with finding effective treatments for depression.
Reduction in Suicidal Thoughts
Another vital benefit of SPRAVATO® is its impact on reducing suicidal ideation. Research indicates that SPRAVATO® can quickly decrease the severity of suicidal thoughts. It provides urgent support for individuals in acute distress and potentially prevents tragic outcomes.
Convenience of Administration
SPRAVATO® is administered as a nasal spray, which many patients find more convenient and less invasive than other forms of treatment. The administration process is straightforward and can be done quickly under the supervision of a healthcare provider, making it a practical option for many.
What to Expect
Treatment sessions are conducted in-office and last 2 hours. A medical provider and technician will monitor you throughout the session, and your vitals will be taken at regular intervals.
There are 2 phases of treatment: Induction and Maintenance.
- Induction indicates the start of your SPRAVATO® treatment, which is as follows: 4 weeks of twice-weekly treatment with at least 1 day in between treatment sessions.
- Maintenance indicates your continued SPRAVATO® treatment and is as follows: once weekly treatment weeks 5 - 8; weeks.
- You and your prescriber determine a 9+ weeks treatment schedule and can be once weekly, once every other week, once every 3 weeks, or an alternative schedule.
The duration of your visit will take place in a private treatment room equipped with a recliner, a guest seating area, and amenities such as a blanket, pillow, fan, and sound machine. After administering the medication under the observation of a technician, you may experience an array of side effects that subside after your treatment session, including but not limited to sedation and feeling like an observer/spectator to yourself (more information in our FAQ section below).
Watch this video in order to learn more about one individual's personal experience with SPRAVATO®
Please read our Full Information SPRAVATO® Pamphlet before submitting a patient interest or Healthcare provider referral form.
Patients must have an active diagnosis of either Treatment Resistant Depression (TRD) or Major Depression with Acute Suicidal Ideation (MDSI) in order to be eligible for treatment with SPRAVATO®.
For more information on SPRAVATO®
Please note the important requirements for treatment in our program before completing the interest form or referral forms:
- Patients must have an active diagnosis of either Treatment Resistant Depression (TRD) or Major Depression with Acute Suicidal Ideation (MDSI) to be eligible for treatment with Spravato.
- Business hours are between 9:00 am – 5:00 pm, with the last treatment appointment scheduled at 3:00 pm.
- Appointments are at least two hours long and require a twice-weekly regimen for the first four weeks, followed by once weekly for one month. Dosing is continued based on response to treatment.
- Patients must have a driver to and from each appointment and are not allowed to resume driving until the next morning.
Patient Interest Form for Spravato Treatment:
For patients who have been seen by an RCBM provider within the past 6 months, please DO NOT complete the forms below. Please discuss your interest with your RCBM provider at your next appointment. Thank you!
If you are a New Patient to RCBM please visit the “New Patient” link on the navigation bar at the top of this page and complete registration. You will select on the registration form that you have interest in Spravato, and the site will automatically route you to complete the Spravato Interest Form.
Open FormHealthcare Professional Referral Form:
Open FormPlease make sure to read the above information. If you have additional questions or concerns, prior to completing the respective interest form, please feel free to email esketamine@rcbm.net.
Doctors answer patients’ top 9 questions about SPRAVATO®.
FAQs
About SPRAVATO®
If you have tried medication and therapy to help with your depression, but none of them helped, SPRAVATO® (esketamine) nasal spray is a new depression medication and the first nasal spray medication specifically for adults with treatment-resistant depression who have tried antidepressants and augmentations but have not had success in symptom relief.
SPRAVATO® (esketamine) nasal spray is indicated for the treatment of:
- Treatment-resistant depression (TRD) in adults as monotherapy or in conjunction with an oral antidepressant.
- Depressive symptoms in adults with major depressive disorder (MDD) with acute suicidal ideation or behavior in conjunction with an oral antidepressant.
The U.S. Food & Drug Administration approved SPRAVATO® on March 5, 2019, and is only available to patients at certified treatment centers in the United States, such as Rochester Center for Behavioral Medicine (RCBM).
This treatment is approved for adults ages 18 and older.
SPRAVATO® is available only through a restricted distribution program called the SPRAVATO® REMS (Risk Evaluation and Mitigation Strategy). A REMS program is in place to ensure the safety of all patients treated with SPRAVATO®. The Rochester Center for Behavioral Medicine’s SPRAVATO® Program operates under the REMS program for service implementation at our office.
The goals of the REMS are to mitigate the risks of serious adverse outcomes resulting from sedation and dissociation caused by SPRAVATO® administration and abuse and misuse of SPRAVATO® by ensuring SPRAVATO® is only dispensed and administered to patients in medically supervised healthcare settings that monitor these patients, ensuring pharmacies and healthcare settings that dispense SPRAVATO® are REMS certified, ensuring patients are informed about serious adverse outcomes from dissociation and sedation and the need for monitoring, and enrolling all patients who receive treatment in an outpatient healthcare setting in a REMS registry to characterize the risks further and support safe use.
Share Link: About SPRAVATO®
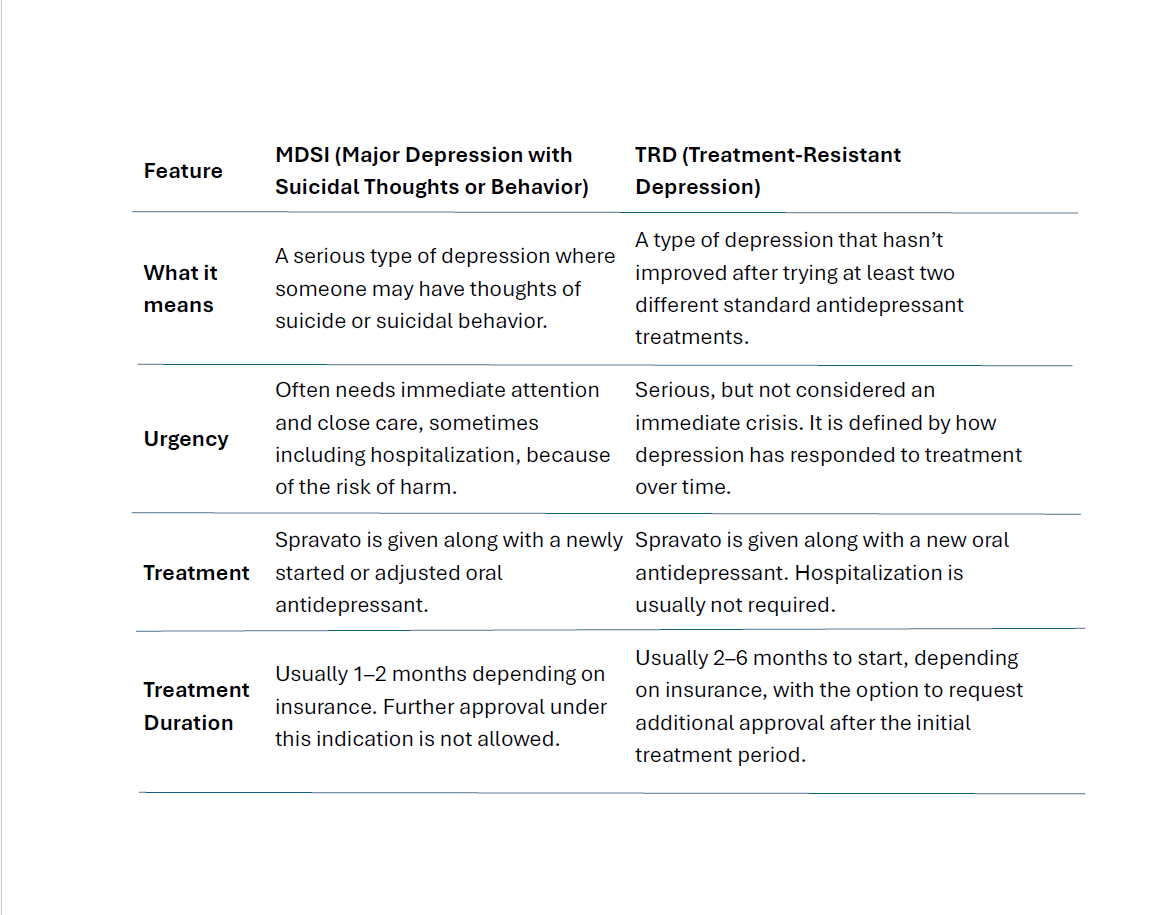
What’s the difference between Major Depression with Suicidal Thoughts or Behavior (MDSI) and Treatment Resistant Depression (TRD) diagnosis?
What is the difference between ketamine and esketamine (SPRAVATO®)?
Ketamine is a generic drug used off-label for depression, while SPRAVATO® is a brand-name nasal spray derived from ketamine and FDA-approved for treatment-resistant depression. While SPRAVATO® and Ketamine are chemically related, SPRAVATO® is not the same as IV Ketamine. Only SPRAVATO® has undergone extensive controlled clinical trials that informed the FDA approval of the medicine for use in adults with treatment-resistant depression and to treat depressive symptoms in adults with major depressive disorder with acute suicidal ideation or behavior.
Esketamine is made from a drug called ketamine, an anesthetic that has also been used for many years to treat depression. Because Esketamine is more potent, it can be used at a lower dose and has fewer side effects. Esketamine has earned FDA approval specifically for use as a nasal spray for those with treatment-resistant depression. Because the FDA approves it, it’s more likely that insurance companies will cover the treatment.
Mathew Perry's tragic story is now widely circulated. As a potential patient, you may have concerns. Please be aware:
- There is a difference between SPRAVATO® and esketamine. SPRAVATO®, the isomer of ketamine, has been approved by the FDA for mental health treatment. Ketamine has not been approved,
- Our post-infusion monitoring adds incredible safety to the medication.
- RCBM has years of experience with SPRAVATO®, dating back to pivotal trials. We studied this medication before it was FDA-approved.
- Our SPRAVATO® dosing is fixed, and toxic blood levels will not be exceeded.
- Mathew Perry's death reminds us of the high stakes associated with depression and treatment and the reason non-mental health experts should not be prescribing this medication.
For more information on Ketamine, please review:
Ketamine for Treatment-Resistant Depression and Related Conditions: A Review of a Novel and Needed Treatment Option by Benjamin Young, MD
Share Link: What is the difference between ketamine and esketamine (SPRAVATO®)?
Who cannot use SPRAVATO®?
You cannot use SPRAVATO® if you…
-Have a Blood Vessel Disease
-Have an abnormal connection between your blood vessels (arteriovenous malformation)
-Have a history of bleeding in the brain (intracerebral hemorrhage)
-Are allergic to Esketamine, Ketamine
-Are Lactating, Pregnant, or planning to become pregnant
-Active substance use disorder
-HIV
Precautions: If you are interested in SPRAVATO®, talk to your healthcare provider if you have the following medical conditions…
- Hypertension (High blood pressure)
- History of Stroke or Heart Attack
- Heart Valve Disease
- History of Brain Injury
- Liver Disease
- History of Psychosis
- History of Seizures
- History of Substance Abuse
- On MAO inhibitors
Share Link: Who cannot use SPRAVATO®?
Does my insurance cover SPRAVATO® treatment?
- Insurance coverage varies based on the individual health plan. We encourage you to check your insurance benefits for SPRAVATO® coverage, including co-pays and deductibles, along with whether SPRAVATO® falls under your medical or pharmacy benefits. The RCBM SPRAVATO® Program does not participate with TriCare, Carelon Behavioral Health, UMR, Beaumont UMR, Blue Care Network, Humana, and Medicaid. If you have one of these health plans, please visit SPRAVATO®.com to find a SPRAVATO® center that takes your insurance.
- All insurances require pre-authorization and continued authorization for SPRAVATO® treatment, while each insurance company has different criteria for authorization of SPRAVATO®. This includes a history of inadequate response to several different classes of anti-depressant medications with a trial of a minimum of 8 - 12 weeks each, along with augmentation strategies.
- Your healthcare provider must refer you for SPRAVATO® treatment and document the medical necessity of SPRAVATO® based on the required criteria.
- If you have a commercial health plan, SPRAVATO with me is available to help you navigate access and affordability.
Share Link: Does my insurance cover SPRAVATO® treatment?
How effective is SPRAVATO® treatment?
In clinical studies, patients who achieved stable remission with SPRAVATO® augmented with an antidepressant showed a 51% decrease in risk of relapse*. In addition, there was a 70% risk reduction for depressive symptoms among those who achieved a stable response compared to an antidepressant alone.
For individuals with TRD
In clinical studies, improvement of depressive symptoms was demonstrated at four weeks by SPRAVATO® plus an oral antidepressant, compared with placebo nasal spray plus an oral antidepressant. Not all patients will respond to SPRAVATO®.
For individuals with MDSI
In clinical studies, those who took SPRAVATO® and an oral antidepressant experienced a greater reduction of depressive symptoms at 24 hours compared to those who took a placebo plus an oral antidepressant. Further reductions were seen consistently through four weeks (25 days) of treatment, so it is essential to follow the treatment plan that you and your healthcare provider have made. Not all patients will respond to SPRAVATO®.
*As of February 2025, the FDA has approved Spravato as monotherapy for adults living with treatment-resistant depression (TRD).
Share Link: How effective is SPRAVATO® treatment?
How does RCBM’s SPRAVATO® Treatment Resistant Depression Clinic stand apart from other programs?
RCBM provides a welcoming and comfortable atmosphere so patients can relax and engage in their treatment. Our staff is highly professional and well-trained. Unlike other centers, our program offers many amenities and services.
Amenities:
- Blankets
- Call buttons for assistance when the technician or medical provider is not present in the room
- Fans and heaters
- Handicap access
- On-site parking
- Pillows
- Power Outlets
- Private treatment rooms
- Recliners
- Snacks and beverages for nausea
- SPRAVATO® playlists on Spotify
- Television in each treatment room
- Waiting Areas for Caregivers
- Wi-Fi
Services:
- Appointment Scheduling
- Appropriate and accurate clinical documentation to ensure continued insurance authorizations so as not to interrupt your treatment
- Communication and coordination of care with your internal and external provider from referral to continued treatment
- Insurance Verification
- Well-trained and attentive staff
Share Link: How does RCBM’s SPRAVATO® Treatment Resistant Depression Clinic stand apart from other programs?
What are common patient questions and answers about SPRAVATO® treatment?
When am I going to begin feeling better? How do I know if it’s working for me?
Patients typically start feeling the benefits of SPRAVATO® around week 8, but the timing can vary from patient to patient. It's important to emphasize that, like oral antidepressants, SPRAVATO® may not be effective for every patient. Patients are encouraged to keep a journal of their feelings during and between SPRAVATO® treatments to assess its effectiveness. This journal can be used to discuss treatment efficacy during follow-up appointments.
How long am I going to have to be treated?
The average length of treatment nationwide is two years. However, each patient's duration is unique and depends on their progress with the medication. The duration is determined based on conversations between the patient and the provider, taking into account the medication's effectiveness and the patient’s ability to maintain a consistent schedule.
How long until I can stop taking additional medications for my depression?
When referring someone for SPRAVATO®, it's important to understand that it is not a cure or a stand-alone medication. It is meant to complement an oral anti-depressant and help with symptom relief. While remission may be possible, it is not guaranteed.
What am I supposed to do during the appointments?
There are no required activities to be done during SPRAVATO®. The only requirement is to administer the medication in order to receive its benefits. However, engaging in activities such as listening to guided therapy/meditation sessions or SPRAVATO® playlists on Spotify may provide additional benefits.
Share Link: What are common patient questions and answers about SPRAVATO® treatment?
Ready to get started? SPRAVATO® referral and evaluation timeline
The following is a timeline of what you should expect from referral through the course of treatment:


Share Link: Ready to get started? SPRAVATO® referral and evaluation timeline